 
The group 18 elements have a full outer shell, while the other partially filled valence shells. How many protons are there in oxygen, carbon, and fluorine? The answer depends on the atom’s composition. The s-shaped electron shell is filled with valence electrons. In other words, the atomic number of the element determines how many electrons it has in the outermost shell. The atomic number is the simplest way to identify which atom has more electrons. The second shell has two subshells, one of which holds two electrons while the other can hold up to eight.Ī molecule is said to have a certain number of electrons in each shell, based on its atomic number. As the first element in this row, Lithium contains three electrons, while neon has ten. The spherical s orbital is the lowest energy shell, while the four higher-energy p orbitals are located in the second shell. The first element in the second row of the periodic table is LithiumLithium, which has three electrons. An atomic number is a useful tool for identifying different elements. 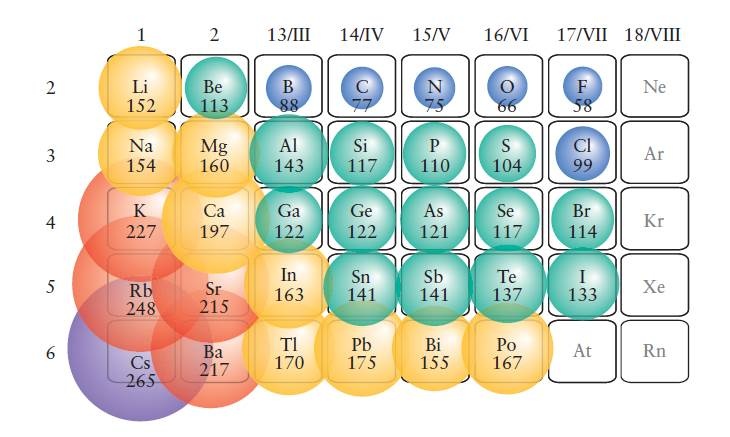
This is due to their atomic numbers, determined by their atomic weight. Elements have two, three, four, or five atomic numbers. The number of electrons in each shell is the atomic number or n. Number of electron shellsĪ chemical element has a certain number of electrons in each shell. However, this trend continues from oxygen to neon, which shows that electronegativity decreases as the number of protons increases. This decrease in atomic radius is due to a positive charge in the nucleus. As electrons are paired with each other, the ionization energy decreases. The outermost shell remains the same during these periods, except for the number of protons. The atomic radius of hydrogen, argon, and oxygen is measured in nanometers. This way, you can find any element’s atomic number and radius. 
The highest number of atoms is hydrogen, and the lowest number is Lithium. Second, you can find the atomic radius of a specific element by using the Periodic Table. There are two basic methods to determine atomic number and radius. Atomic radiusįirst, you need to know the atomic number of the element. In addition, the Bohr model makes use of different electron shells that have a specific number of electrons. Each of these atoms has two electron shells, the outermost of which contains the valence electron. The Bohr model of oxygen-carbon fluorine neon li is a visual representation of electrons orbiting a small nucleus. Final Words What Is The Bohr Model Of Oxygen Carbon Flourine Neon Lithium?.The Bohr Model of Oxygen Carbon Fluorine Neon Lithium.What Is The Bohr Model Of Oxygen Carbon Flourine Neon Lithium?. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
